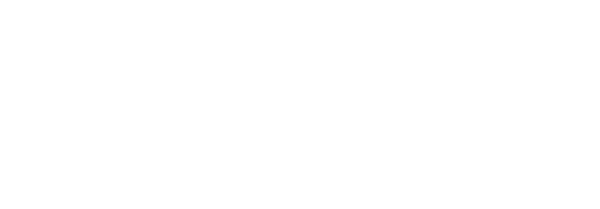Ways agricultural pesticides can contaminate water

The content of this article 'Ways agricultural pesticides can contaminate water' was prepared by Douglass Stevenson, Paul Baumann, John Jackman of the Texas A&M AgriLife Extension and has been revised and republished by FreshFruitPortal.com.
The over-application or misuse of pesticides and other agricultural chemicals (such as fertilizers) can allow these chemicals to enter surface and ground water.
Drift, evaporation and wind erosion can carry pesticide residues into the atmosphere. From there they can fall in rain or snow to contaminate lakes and streams.
Using excessive amounts of chemicals on open or porous soils where there are shallow water tables can allow pesticides to leach or percolate into the ground water.
Improperly cleaning or disposing of containers, as well as mixing and loading pesticides in areas where residues or run-off are likely to threaten surface or ground water, are other potential sources of contamination.
Some pesticide labels and some state statutes specify safe distances from well heads for pesticide mixing and loading.
Agricultural chemicals also can pollute surface water through irrigation return flow and rainfall runoff.
Carefully following label directions about proper dosage and application methods can greatly reduce the possibility of water contamination.
Pesticide Properties
Properties that affect a pesticide’s potential to pollute water include formulation, toxicity, persistence, volatility, solubility in water, and soil adsorption.
Of course, pollution risk also is affected by soil characteristics, application methods, weather and other factors.
Formulation
Pesticides come in several physical forms or formulations that make them easy to store, transport and apply, and that help in controlling target pests.
Common formulations include water dispersible granules, wettable powders, dusts, aerosols, solid or liquid baits, granules, emulsifiable and flowable concentrates and solutions.
There are other less common formulations designed to give special properties to the pesticide mixture or to take advantage of properties of active ingredients or protect the environment.
These include microcapsules, plastic beads, plastic membranes, plastic ropes, controlled release dispensers and others.
While most environmental hazards come from the active ingredient in a pesticide, the way its formulation interacts with the environment determines the overall hazard of a pesticide.
Spray formulations can drift with the wind or vaporize into the air.
Other formulations can leach into ground water or be carried into surface water by rainfall or irrigation runoff.
Even pesticides in formulations that bind them to soil particles can find their way into surface waters if soil is eroded by wind or water.
Toxicity of pesticides
The active ingredient is the chemical compound in a pesticide that kills or otherwise affects the target pest.
Other substances in a pesticide formulation are inert ingredients that act as carriers and preservatives for active ingredients, and also make mixing and application easier.
When determining whether and how to register a pesticide, the EPA considers the toxicity of the active ingredient.
Toxicity is determined by the amount required to produce biological effects.
Dose and Effective Dose
A dose is the amount of a substance used at one time. Most substances are toxic at large enough doses, but harmless or even beneficial at lower doses.
Drinking water is an example. People need to drink some water every day. However, drinking the equivalent of 15 percent of one’s body weight can be fatal.
Similarly, table salt is absolutely necessary for proper health, but as little as 1 ounce (2 Tablespoons) of table salt would deliver a lethal dose to a 1-year old child.
There is a lethal dose of caffeine in 100 cups of coffee. There is a lethal dose of oxalic acid in 20 pounds of spinach.
There is a lethal dose of aspirin in 100 tablets. We can compare aspirin with two chemical pesticides. Malathion is about half as toxic as aspirin. Parathion is 70 times more toxic than aspirin.
The hazards of pesticide residues are negligible compared to the dangers from common household chemicals and medicines.
Table 2 compares toxicities of common products with pesticides.
The effective dose is the amount of a substance needed to kill or otherwise affect a target pest. Amounts less than the effective dose will likely not kill the target pest.
Amounts greater than the effective dose will not necessarily be more effective in killing the target pest.
Instead, this larger dose may kill more non-target organisms, cost more, and pollute the environment. Common measures of a chemical’s toxicity are the LD50 and LC50.
These measures refer to doses that kill 50 percent of the animals in a test group. These toxicity terms can apply to target pests or non-target organisms, including humans.
The toxicity of a substance determines its proper dosage.
The LD50 is the dose of a particular material, taken through the mouth, skin, or inhaled, that is lethal to 50 percent of a group of test animals.
The higher an LD50 is, the lower the toxicity of the substance. Items with low LD50s are extremely toxic.
Basic measuring units used are milligrams of toxin per kilograms of body weight, or “mg/kg.”
Table 2 shows the LD50 values in rats for various pesticides and other familiar chemicals.
Aspirin, table salt and other common natural products provide comparisons. EPA uses LD50s to determine the safe level of pesticide residues in water.
LC50 is another measure of toxicity. LC50 stands for the concentration of a material in air or water that will kill 50 percent of the animals tested.
The toxicity of a pesticide is different from the hazard it represents.
Hazard refers to the likelihood that a substance will cause harm under certain conditions.
For example, the pesticide paraquat is highly toxic. Just a few drops can kill an adult human. There is no antidote for paraquat poisoning.
Used properly and stored in a tight container, paraquat has high toxicity and a low hazard.
If the contents of the container spill, however, the toxicity remains the same but the hazard increases enormously.